Methanol & Ammonia Tell A Good Shipping Fuel Story, But Stop Before The Tragic Finale
Support CleanTechnica's work through a Substack subscription or on Stripe.
Imagine, if you will, a sack of fertilizer and a flask of wood alcohol, sitting at a bar. Through the entrance comes a streamlined ship of the sea, a proud and beautiful cargo cruiser of the waterways of the world. She’s come to drink away her sorrows at her long-standing relationship with the bad boy of energy, residual bunker fuel, known by his friends and enemies simply as resid.
Both the sack and the flask immediately set out to woo the vessel. And so they tell her their stories of how much better they would treat her and her sisters than resid would, how responsible they are, how long their own histories are. They tell her that while they are traditionally do not consort with beautiful marine vessels like her, instead having long-standing, non-monogamous relationships with fields and industrial plants, they would like to welcome her and her sisters into their non-traditional relationship.
Now, both fertilizer and flask have dirty secrets of their own. Both spring from the same womb as resid. Every ton of them is made from crude oil’s siblings natural gas and coal today, and while natural gas had a much better image once, it has since been found to be almost as coarse as resid beneath the dapper attire.
But they both promise that they are working on being better, that they will clean up their ways, that they have big plans to improve themselves, and besides, compared to resid they are practically altar boys. They both tell her that they are working on parting ways with gas and coal, and instead turning to water, specifically the squeakiest clean part of water, the saintly hydrogen. Apply a little tingle of electricity, and the saint is released to be turned into the contents of the flask and sack. If not hydrogen, then otherwise unused leafy green plant material.
It’s working, too. The ship is leaning toward them, hoping to be able to get free of the cruel grip of resid and the abuse he wreaks everywhere he goes. She’s willing to believe their stories, but somewhere in the back of her mind, a little voice is asking, “Is this too good to be true?”
Of course it is.
When I was in Glasgow at Stena Sphere’s technical summit recently, methanol as a shipping fuel was central to the discussions of maritime decarbonization. Ammonia was smelling up the wings but not present, as an ammonia organization had declined to send a representative. Stena has not ruled out ammonia, but does have a joint venture with Proman for methanol, which means that stakeholders in the group are already somewhat committed to the pathway. That’s going to be challenge for them, but Maersk is much more committed.
Let’s start with the basics. Methanol is an alcohol that is manufactured from natural gas right now. My sources suggest 170 million tons of annual market, but the Proman representative Peter Schild used 120 million tons as the figure. Regardless, between 100 and 200 million tons of methanol is already manufactured around the world. The 800-pound gorilla, Methanex, buys old chemical processing plants, rejigs them for methanol manufacturing relatively close to markets and ships short distances to customers. The methanol industry loves the idea that its product will become the clean fuel for maritime shipping, as even with my 170 million tons and massive electrification and efficiency programs cutting burnable fuel demands by two-thirds, what’s left over is a tripling of their annual market. That’s not a new market, that’s a multiplication of their market. If the methanol industry can make their case, they’ll be ramping up plants globally and all the current major players will be giving their shareholders dividends hard to dream of before the transition.
Ammonia isn’t an alcohol. It’s a nitrogen atom and three hydrogen atoms. We manufacture about 150 million tons of it as well, almost entirely for fertilizer, and also manufacture it mostly from natural gas. It’s not as good at burning as methanol, it has the advantage that it doesn’t have carbon, so no CO2, and the disadvantage that it has lots of nitrogen and so is more likely to create N2O with its global warming potential of 265 times that of CO2. Oh, and when it interacts with water it turns from a liquid whose fumes will screw a sailor up for life to a corrosive gas that will just kill them before turning into a third chemical that goes back to being really bad for human health.
Neither ammonia or methanol can share tanks with other fuels. They are other. They are separate. So in ports, if we wanted to use them as marine fuels, we’d need new bunkering wharfs, barges and tankers, and new pumps and hoses — not an insurmountable requirement. We ship both of them around the world, as both are essential industrial and agricultural feedstocks. Most ports aren’t equipped to deal with either of them. While the Proman representative’s slides talked about 122 ports with methanol storage, there are about 800 ports worthy of the name and the ports with methanol storage aren’t focused on putting the methanol in fuel tanks, but distribution storage tanks.
As both ammonia and methanol are made from fossil fuels, they are both climate change problems.
Methanol is made from a process that turns the natural gas (55%), coal (35%), or other fossil gases (10%) into an intermediate synthetic gas which it then distills into alcohol. While Methanex makes the claim that its manufacturing carbon footprint is 0.2 tons of CO2e per ton of methanol, the industries own makes it clear that average best case scenario for end-to-end natural gas methanol when used as a fuel is 2 tons of CO2e per ton of methanol.
And that best case matters. The Methanol Institute’s guidance to the various modeling agencies they employed were to use EU standards for upstream methane emissions, and the EU is best of breed on that. The USA’s emissions have been found to be significantly understated by the EPA, and are in the range of four times that of the EU. At US levels of upstream emissions from natural gas, end-to-end methanol as a fuel is more like 3 tons of CO2e per ton of methanol.
And the coal methanol is, unsurprisingly, worse at about 6 tons of CO2e per ton of methanol.
The average of all of this is about 4 tons of CO2e per ton of methanol.
And then there’s the kicker. Methanol has about 45% the energy density by mass as resid or diesel. That 4 tons becomes 8.9 tons. Yeah, burning methanol instead of more directly burning fossil fuels results in 2.9 times the CO2e emissions for the same distances traveled. In its current form, methanol isn’t remotely a solution, it’s an amplification of the problem by a factor of three.
Ammonia represents demand for about 30 million tons of hydrogen a year, and each ton of hydrogen drags 8-10 tons of CO2e behind it between upstream emissions and CO2 from steam reformation with natural gas. That’s about 7 tons of CO2e per ton of ammonia. When ammonia is put on fields, part of it turns into N2O, one of the oxides of nitrogen, with its high GWP of 265 times that of CO2. That means the lowest emissions form of ammonia’s dominant use results in about 10 tons of CO2e per ton of fertilizer. It’s a big climate change problem, in the same magnitude range as global aviation and marine shipping, and requires fixing.
Oh, but wait. This begs the question. If putting it on fields produces lots of nitrous oxides, what does burning it do? After all, any combustion in our nitrogen-rich atmosphere tends to bind oxygen and nitrogen into N2O. Of course, this has been studied. The range of combustion in maritime engines is from 0.06 grams of N2O per kWh to 1.95 grams of N2O per kWh. What does that do to the already high tons of CO2e per ton of ammonia?
Ammonia has an energy density of 15.6 MJ/L. That 15.6 MJ is 4.3 kWh. Burning it in a high-efficiency marine engine means that we would get about 50% of that energy as useful forward motion, or 2.16 kWh to be generous. Heading up to metric tons, that means about 2,160 kWh. At 0.06 grams of N2O per kWh, that’s about 0.000132 tons of N2O. At a GWP of 265, that’s the equivalent of only 0.03 tons of CO2e per ton of ammonia. That’s not bad.
But the increments of emissions go up fast. The next one is 0.47 grams N2O per kWh. Plugging that in gets 0.26 tons of CO2e per ton of ammonia on top of the existing CO2e debt. The top end of ammonia emissions is 1.95 grams of N2O per kWh and plugging that in results in 1.1 tons CO2e per ton of ammonia.
Achieving the best case scenario requires engines carefully designed to create those levels, careful maintenance, and no contaminants in the ammonia. For this workup, we’ll assume that on average the second lowest is most likely, and add 0.26 tons of CO2e to emissions.
And it has a lower energy density than methanol, about 42% that of resid or diesel. That means that while it doesn’t produce CO2 when burned (no carbon in the NH3 molecule), its actual carbon footprint per nautical mile is over six times that of resid or diesel today.
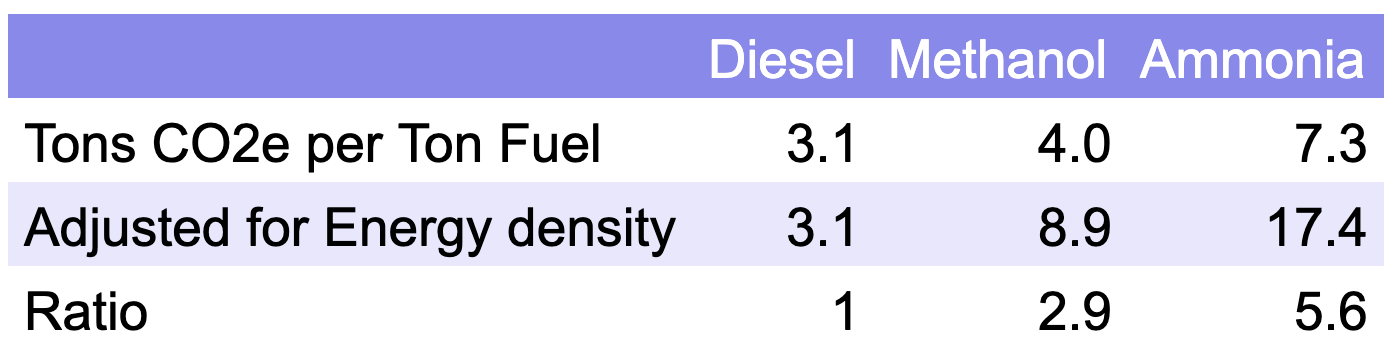
Unabated methanol used today in marine engines would result in full-lifecycle greenhouse gas emissions 2.9 times that of burning current maritime fossils fuels. It’s much worse for ammonia, with 6.4 times the full-lifecycle emissions.
And don’t forget that an ammonia spill in a major port could result in tens of thousands of human deaths, and an ammonia leak in the enclosed spaces of a ship could kill multiple sailors. Frankly, I was baffled before doing this work-up about why anyone would consider ammonia, but it’s still going.
Given these greenhouse gas emissions, obviously they must have a solution, and of course, they do. Mostly it’s a green hydrogen story, but there’s a side story for methanol that might make it viable, so let’s start there.
Natural gas is about 88% to 92% methane (CH4) and various trace elements. As I’ve been noting, most recently in my biofuels pathways survey, we unintentionally create a lot of methane from how our agriculture and waste streams are organized. Every pile of rotting agricultural stalks, livestock dung, and every landfill site with food waste in it is rotting beneath its surface without any oxygen around. Microbes that are fine with that eat the biomass and emit methane.
As we are dumping a full third of our food production as waste, about 2.5 billion tons a year, as 20% of land mass is under crops where we mostly only use the fruits, and as 20% or so is devoted to animal husbandry, that’s an awful lot of biomethane produced every year. I don’t know how much, but methane has a direct global warming forcing that’s 25-72 times that of CO2, so any emissions are a problem. Biomethane degrades back into mostly CO2, which reduces the indirect forcing a bit, but 25-72x the GWP of CO2 is still bad.
Some of that biomethane is concentrated or can be concentrated. Landfills can and are drilled with collection pipes. Cattle milking barns can have the methane filtered from the ventilation system. Most of it isn’t concentrated today, but it is possible to capture at least some of the methane emissions we are causing. And if we do that and put it into existing methanol production processes, it behaves exactly the same way as natural gas, and so we take a climate change problem on the one hand (biomethane) and use it to eliminate a climate change problem on the other hand (fossil methanol).
Of course, that’s not how reality works. In reality, the people promoting biomethane are selling anaerobic digesters which much more quickly create methane from rotting biomass. In my world, where I’m looking for actual climate solutions, intentionally making more methane in a leaking distribution and use system isn’t a climate solution, but a problem. I’ve thought through the logistics of this, and while it’s possible to make a relatively low-emission end-to-end anaerobic digestion and integrated methanol plant, it requires a lot of careful design and ongoing effort at every step of the process to prevent anaerobic digestion in the accumulating feedstocks and emissions from the production. And to be clear, some of this is shared with my preferred pathway of biofuels that don’t involve biomethane. Bringing large amounts of biomass to an industrial biofuels facility will require some care to reduce methane emissions in transit and storage.
It’s like comparing Norway’s high-quality methane leakage control and monitoring to the USA’s, Russia’s, and the Middle East’s abysmal track records. There will be leading practices, but they will cost money and careful attention, and the vast majority of the producers won’t spend the money or take the care.
Anaerobic digesters, in other words, are as much a climate change problem as a climate change solution. I’m not a fan. This makes Maersk’s planned 50,000 tons of biomethane-sourced methanol expected delivery in 2024 probably better than fossil methane, but a lot depends on the quality of the containment of the biomethane created and used, and care in management of the feedstock so that it doesn’t just rot anaerobically while while to be shoved into the anaerobic digesters.
And the other option, bringing the methanol reactor to the source of methane we are creating, doesn’t cut it either. Paul Martin is in the business of making chemical processing plants like those, and has been for decades. He reminded me that this kind of thing has been tried before, with modular small chemical processing plants attached to an oil field’s methane flaring system, diverting the completely free methane into a small plant to make methanol or other substances. Despite completely free feedstock that’s already concentrated, none of these plants could make any money. That was against the price point of unabated methanol and similar substances, so perhaps there’s some faint hope today, but chemical processing likes to be scaled, with multiple as-big-as-possible modular chemical plants consolidated on the same site, with strong efficiencies of supply and distribution.
I’m still very bullish on reducing or mitigating biomethane production at source. That includes seaweed extract food supplements for livestock that reduce methane belching by up to 80%, burning or generating electricity from landfill methane emissions at the site, mechanically rotating plant matter middens to allow decomposition that produces CO2 instead of methane, clearing biomass rigorously from hydro reservoirs before filling, and diverting as much biomass waste as possible into mostly non-methanol pathways for biofuels.
Setting aside biomethane to methanol, both methanol and ammonia have another pathway: green hydrogen.
This is more problematic for methanol than ammonia, which is part of ammonia’s pitch. Ammonia plants have a natural gas pipeline going in, they use steam reformation to split the methane into hydrogen and CO2, they dump the CO2 into the air and put the hydrogen into their process, which binds it with nitrogen from the air. Replace the natural gas pipe and steam reformation component with a water pipe and an electricity line, and replace the steam reformation component with an electrolyzer, and it’s business as usual after that.
Most of the balance of plant can be maintained. That’s great because it means those roughly 7 tons of CO2e per ton of ammonia have a clear pathway to the future. Don’t forget, they are a big consumer of hydrogen to make their 150 million tons of ammonia today. They have a pathway to fixing their primary climate problem, but mostly the ammonia industry is focused on multiplying the climate problem by selling ammonia as a shipping fuel.
To be clear, if the ships were powered by green ammonia made only with low-carbon electricity, that would be a climate win, but every joule of energy in ammonia is hydrogen. And turning water into hydrogen with electricity, even at point of consumption, throws away 30% to 40% of the energy in the electricity. This is reasonable when the hydrogen isn’t the point, the nitrogen is, and it’s used as a fertilizer to feed our teeming billions, but it’s not economically reasonable for fuels.
Methanol can be made synthetically by starting with hydrogen and carbon molecules, but that’s not how it’s made today. While the claim is that they will be able to adapt their plants, it’s a completely different technology with different feedstocks. They will be able to preserve the shell of the building, the methanol storage tanks, and the distribution network, but since they are located where there was lots of natural gas or coal, not lots of electricity and CO2, most of those plants aren’t in the right places. That’s a problem that’s somewhat shared by ammonia, which gets its nitrogen from the air, but still needs lots of water and electricity it might not be well sited for.
The biomethanol pathway has similar problems, as the plants are once again located where there is a lot of natural gas and coal, not good feedstocks for anaerobic digesters. The output of the digesters would preserve the methanol manufacturing facility, but add capital costs and operational costs.
Both of the replacements with green hydrogen as a feedstock require significant capital cost expenditures between MW++ electricity at the plants, electrolyzers, and water deliveries. It looks like methanol’s would be higher given complete process replacement. As I’ve noted time and again, hydrogen can be green, but it can’t be cheap. They both have two choices. The first is that they can scale the hydrogen electrolyzer to produce the amount of hydrogen that they require for continuous operation, which requires firmed electricity which will cost more. Or they can scale the electrolyzer to produce hydrogen when electricity from renewables happens to be operating, which will mean that the electrolysis plant capex will be a significantly bigger portion of the cost case, and they’ll have to include a bunch of hydrogen storage and compression in their capital and operating costs.
The net result is that while methanol and ammonia are relatively inexpensive industrial commodities when made with unabated natural gas while using the atmosphere as an open sewer, they are going to be much more expensive in the future. And methanol at least is already more expensive than diesel in most of the world, up to 2.2 times as expensive for the same energy. And ammonia costs more than methanol does while having even less energy per ton.
And so back to the story. The claims that the methanol and ammonia industries are making are seductive. They state, rightly, that they are globally-scaled industries today producing high-quality methanol and ammonia that ships can be used in the appropriate engines today. They state rightly that while more expensive than diesel or resid today, they also burn more cleanly outside of CO2e emissions, with no sulphur or carbon black emissions. They state rightly that they have obvious pathways to having much lower carbon emissions. They claim, less correctly, that they can reuse existing infrastructure and processes. They claim, rightly, that they are a bit cheaper today than biodiesel.
But what they leave out of their stories — and remember I listened to the Proman methanol pitch personally recently — is that using current methanol or ammonia that’s been made from natural gas would be a climate disaster and that the maritime industry investing in capital expenditures in ports and on ships to use methanol and ammonia are locking themselves into vastly more expensive future operating costs for fuel. And they leave out the reality that biodiesel is going to get a lot cheaper as it scales massively.
When listening to sales pitches, it’s good to question the numbers you are hearing and to ask yourself what the salesperson is omitting. In sales pitches for methanol and ammonia for maritime fuels, the numbers don’t add up, and the omissions are glaring. The more I dig into them, the more I’m comfortable that batteries and biodiesel will dominate.
Mea culpa: an earlier version of this article misstated the emissions of methanol, because oddly, the Methanol Institute’s own publication misquoted their body in the executive summary, asserting that the emissions that they were counting were only for manufacturing and didn’t include end use as a fuel. Paul Martin spotted that mistake. Others questioned my CO2 emissions from burning methanol. As a result I pulled the article until I had the opportunity to recalculate the actual emissions. That led me to assess the provenance of the institute’s claims on natural gas upstream emissions and the percentages from coal. The net result was a lowering of current methanol’s emissions if used as a maritime alternative fuel from hideously bad end-to-end to merely horribly bad.
Sign up for CleanTechnica's Weekly Substack for Zach and Scott's in-depth analyses and high level summaries, sign up for our daily newsletter, and follow us on Google News!
Have a tip for CleanTechnica? Want to advertise? Want to suggest a guest for our CleanTech Talk podcast? Contact us here.
Sign up for our daily newsletter for 15 new cleantech stories a day. Or sign up for our weekly one on top stories of the week if daily is too frequent.
CleanTechnica uses affiliate links. See our policy here.
CleanTechnica's Comment Policy

